Racemic modification refers to the process of creating a racemic mixture, which is an equimolar mixture of two enantiomers (mirror-image isomers) of a chiral compound. A racemic mixture shows no optical activity because the optical rotations of the enantiomers cancel each other out. On the other hand, the resolution of a racemic mixture involves separating the two enantiomers from each other, which is a fundamental task in stereochemistry, especially in industries like pharmaceuticals, where the biological activity of enantiomers can differ significantly.
This note will cover the following aspects in detail:
- Racemic modification.
- Physical and chemical properties of racemic mixtures.
- Methods of racemic modification.
- Resolution of racemic mixtures.
- Methods of resolution.
- Industrial and pharmaceutical significance.
Racemic Modification
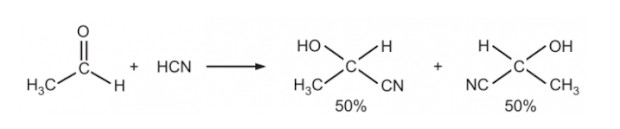
Racemic modification is the process by which a chiral substance (that has one enantiomeric form) is converted into a racemic mixture. It is the formation of an equal mixture of two enantiomers, typically due to the loss of stereoselectivity in a reaction. The mixture contains both the R and S forms of the molecule in equal proportions.
Mechanisms of Racemic Modification
- Synthesis from Achiral Precursors: During the synthesis of chiral molecules from achiral precursors, a racemic mixture is often formed because the reaction has no preference for creating one enantiomer over the other.
- Racemization: A single enantiomer can convert to its racemic form through a process called racemization. This occurs when the chiral center becomes unstable and interconverts between its two possible configurations. Racemization can be induced by heat, light, or catalytic processes.
Examples of Racemic Modification:
- SN1 Reactions: In an SN1 reaction, a planar carbocation intermediate is formed, which can be attacked by a nucleophile from either side, leading to equal amounts of both enantiomers, forming a racemic mixture.
- Protonation of Amines: Chiral amines can undergo racemization in the presence of acidic conditions, leading to racemic mixtures.
Physical and Chemical Properties of Racemic Mixtures
The properties of a racemic mixture are often different from those of the individual enantiomers:
- Optical Activity: While individual enantiomers are optically active (they rotate plane-polarized light), a racemic mixture shows no net optical rotation because the effects of the two enantiomers cancel out.
- Melting and Boiling Points: Racemic mixtures often have different melting and boiling points compared to their pure enantiomers. For example, a racemic mixture may have a lower melting point (called racemic melting point depression) than the individual enantiomers.
- Crystalline Forms: Racemic mixtures can crystallize differently compared to pure enantiomers. In some cases, they form racemic crystals (where both enantiomers are present in the same crystal structure) or conglomerates (where the two enantiomers crystallize separately).
Methods of Racemic Modification
Racemic modification typically involves non-stereoselective reactions or racemization processes that can be controlled or induced.
a. Non-stereoselective Synthesis
Many organic reactions that produce chiral centers from achiral starting materials are non-stereoselective. For example, the addition of a nucleophile to a carbonyl group may form a new chiral center, but since there is no preference for one enantiomer over the other, a racemic mixture is often the result.
b. Racemization
Racemization occurs when one enantiomer spontaneously converts into the other, typically through an intermediate that does not have a chiral center. Common methods of racemization include:
- Thermal racemization: Heating can promote racemization, particularly in compounds with labile stereocenters.
- Acid/base catalysis: Acidic or basic conditions can promote racemization by temporarily removing the chirality of the molecule (e.g., by forming an achiral intermediate like a planar carbocation or enolate).
Resolution of Racemic Mixtures
The resolution of racemic mixtures is the process of separating the two enantiomers that constitute a racemic mixture. Since enantiomers have identical physical and chemical properties in achiral environments, they cannot be separated by traditional methods like distillation or recrystallization based solely on boiling or melting points. Instead, resolution methods rely on exploiting the different behaviors of enantiomers in chiral environments.
Why is Resolution Important?
- Pharmaceuticals: Many drugs are chiral, and often only one enantiomer is therapeutically active or safer. Separating enantiomers allows for the production of pure, effective drugs.
- Asymmetric Synthesis: Although asymmetric synthesis aims to directly produce only one enantiomer, resolution is often needed to separate racemic mixtures obtained from traditional synthetic routes.
Methods of Resolution
There are several methods to resolve racemic mixtures into their individual enantiomers:
a. Mechanical Resolution
In this rare and labor-intensive method, conglomerates (mixtures of enantiomerically pure crystals) are physically separated by hand or mechanical means. This method is historically significant but impractical for most modern applications.
- Example: Louis Pasteur first achieved the mechanical resolution of racemic tartaric acid by manually separating the rightand left-handed crystals under a microscope.
b. Chemical Resolution Using Chiral Resolving Agents
One of the most common methods of resolution involves converting the racemic mixture into diastereomers, which have different physical and chemical properties and can be separated by conventional methods like crystallization or chromatography.
- Steps:
- React the racemic mixture with a chiral resolving agent to form diastereomeric salts or derivatives.
- Separate the diastereomers through crystallization or chromatography.
- Regenerate the pure enantiomers by removing the chiral resolving agent.
- Example: Tartaric acid is frequently used as a chiral resolving agent to separate enantiomers of amines by forming diastereomeric salts.
c. Chiral Chromatography
In chiral chromatography, a racemic mixture is passed through a chiral stationary phase (such as a chiral column in high-performance liquid chromatography, HPLC), which interacts differently with each enantiomer. This results in different retention times for the enantiomers, allowing them to be separated.
- Example: Chiral HPLC is widely used in the pharmaceutical industry to resolve enantiomers of chiral drugs.
d. Enzymatic Resolution
Enzymes, which are inherently chiral, can selectively react with one enantiomer of a racemic mixture, leaving the other enantiomer unchanged. This method is highly specific and is commonly used in the resolution of alcohols, esters, and amines.
- Example: Lipases can hydrolyze only one enantiomer in a racemic ester, leaving the other enantiomer intact. This process is valuable in industrial-scale resolutions of racemic mixtures.
e. Kinetic Resolution
In kinetic resolution, the enantiomers in a racemic mixture react at different rates with a chiral reagent or catalyst. One enantiomer reacts faster and is consumed, while the slower-reacting enantiomer remains, allowing for their separation.
- Example: The Sharpless epoxidation is a common asymmetric reaction where one enantiomer is preferentially epoxidized, leaving the other enantiomer behind.
f. Chiral Auxiliaries
A chiral auxiliary is a temporary chiral group that is attached to the racemic substrate, making the two enantiomers behave differently during subsequent reactions. After the reaction, the auxiliary is removed, yielding the enantiomerically pure product.
- Example: The Evans auxiliary is a widely used chiral auxiliary that induces diastereoselectivity in various organic reactions, leading to the formation of enantiomerically pure compounds after removal of the auxiliary.
Industrial and Pharmaceutical Significance
a. Pharmaceutical Applications
The resolution of racemic mixtures is particularly important in the pharmaceutical industry, where the biological activity of enantiomers can differ drastically. A drug’s efficacy and safety can depend on the enantiomer used. For instance, S-thalidomide caused severe birth defects, while R-thalidomide had sedative properties. This led to strict regulations requiring the separation and testing of individual enantiomers of drugs.
b. Asymmetric Synthesis vs. Resolution
While asymmetric synthesis aims to produce only the desired enantiomer from the start, resolution of racemic mixtures is still widely used due to the simplicity of certain synthetic methods that produce racemates. However, the increasing focus on green chemistry and more efficient production processes is pushing the industry toward the development of enantioselective catalysts and asymmetric synthesis methods.
Conclusion
Racemic modification is the process of creating a racemic mixture, while resolution involves separating the individual enantiomers from that mixture. Resolution is crucial in many industries, particularly pharmaceuticals, where enantiomers can have different biological effects. Several methods, including mechanical resolution, chemical resolution, chiral chromatography, enzymatic resolution, and kinetic resolution, are employed to achieve the separation of enantiomers. The importance of these processes continues to grow as stereochemistry plays an increasingly critical role in drug development, chemical synthesis, and biological applications.

